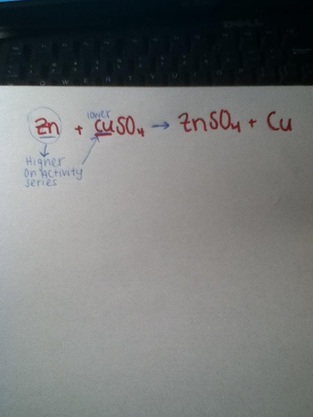Zinc Sulfate
Gabrielle Benda
Period: 2
1/3/13
Abstract: Zinc Sulfate is an inorganic compound used to protect agriculture, provide the manufacturing of rayon, and helps zinc deficiency. Zinc was discovered in Germany in 1746 by Andreas Marggraf. Years later, Zinc Sulfate was produced by treating Zinc with an aqueous sulfuric acid. Zinc Sulfate's melting point is 212 degrees Farenheit, and its boiling point is 952 degrees Farenheit. The chemical reaction is as follows: Zn + H2SO4 + 7H20 ------ ZnSO4 (H20)7 + H2. Zinc Sulfate is 100% soluable in water and its pH level is 5, which makes it a weak acid. In order to produce ZnSO4 you need the chemicals Zinc and Copper Sulfate (II). During a single replacement reaction Zinc combines with Sulfate making ZnSO4 and Copper becomes independent. Once the chemicals react, copper sits at the bottom of the beaker changing into an average copper color because the Sulfate has combined with the Zinc.
Zinc Sulfate
Zinc sulfate is an inorganic compound with the formula of ZnSO4. It is known as
“white vitriol”, which is a colorless and odorless water-soluble white powder or
crystalline structure. It is not flammable or explosive, but it will decompose
in extreme heat. (1) Zinc sulfate’s melting point is 212F, and its boiling
point is 952F. Zinc sulfate is also 100% soluble in water, and its pH level is
5. The element zinc was discovered in Germany in 1746 by Andreas Marggraf.
Marggraf had produced zinc in 1746 by heating a mixture of calamine
and carbon without copper. However, the development of zinc had already been
created by a man in England, but Marggraf received credit because of his detail
in the experiment.(6) Many years later, zinc sulfate was produced by treating zinc with an aqueous
sulfuric acid. The chemical reaction is as follows: Zn + H2SO4 + 7 H2O → ZnSO4
(H2O) 7 + H2.(5) Zinc sulfate is used in the medical field
to treat a low zinc level, or zinc deficiency. (2) It is also used to provide
the required zinc amounts in animal feeds. (4) It is used in fertilizers to provide a
source of zinc for crops and plants. It's used as an ingredient in the
production of rayon fiber, and as an ingredient in flame-proof material. In some
cases, it can replace zinc oxide, depending on its use. Zinc sulfate is used in
industry, agriculture, and in medical uses. (1)
Zinc is used in the manufacturing process for making rayon. Rayon is a
textile fiber made from regenerated cellulose. A solution of zinc sulfate,
sulfuric acid, and sodium are injected into raw rayon to help make tires. Also
when, zinc sulfate and sulfuric acid combine it can produce electricity. Another
way the element is effective is you can use it to help remove moss stains on
roofs of homes. (1)
Zinc sulfate is also an important element in agriculture. It is used to
protect a wide range of food products containing wheat and cotton. The element
also protects pears, cabbage, apples, and broccoli from fungal infestation when
a solution of zinc sulfate is sprayed on them. It also is used to provide the required
zinc amounts in animal feeds, and fertilizers, as stated above. Almost all
fertilizers use zinc sulfate because of its positive benefit to plants. (4)
Zinc sulfate is most commonly prescribed to help with zinc deficiency and
to aid keeping body tissues healthy. (2) The element is used as a dietary
supplement when people have low zinc in their system. It also protects the skin
when used in creams for treatment of acne, or as sunburn protection, and as an
aid for after the skin burns. Both over-the-counter and prescription acne
medicines contain zinc sulfate, as well as other skin care products, because of
its ability to kill microorganisms on the skin. (1) Zinc sulfate is a very
effective method for treating zinc deficiency, causing a person's zinc level to
rise significantly when taken as a supplement. (4)
Though zinc sulfate can be useful for many purposes, it can be a
dangerous irritant and pollutant if used or handled incorrectly. If contact with
zinc sulfate is made with eyes or skin, severe irritation may occur. If inhaled,
it will cause irritation of the throat and nose. (3) Medical attention may be
required for any of these situations. Because zinc sulfate can be harmful if used or handled improperly, it is important to
take the necessary steps to avoid such occurrences. (6) Zinc sulfate is most
commonly found as a water-soluble powder. (1) To avoid inhaling this powder,
wear a face mask that covers both the nose and mouth. Wearing gloves, goggles,
and a mask is also a safe bet when mixing or handling zinc sulfate in its liquid
form. If it comes into contact with your skin, it could result in itchiness.
If dusty it may cause breathing difficulty and irritation of mucous membranes. (3) Ingestion may cause strong
stomach cramps and diarrhea and may cause vomiting. (6) Chronic health hazards
include stomach irritation, abdominal cramps and nausea. (5) This product is
also highly soluble in water and has the potential to be toxic to fish and other
aquatic life. It also has the potential to be toxic to plant life and other
organisms living in soils. (1)
Overall the element zinc sulfate has many benefits. It helps the
manufacturing process with producing rayon fiber for tires. The element protects
foods from fungal infestation. Lastly, zinc sulfate helps people keep their body
tissues healthy. Many living organisms are dependent on zinc sulfate and this is
why the element is so significant. (1)
***Procedure***
Zn+ CuSO4 -------ZnSO4 + Cu
The Balanced Equation above is a Single Replacement Reaction
Procedures:
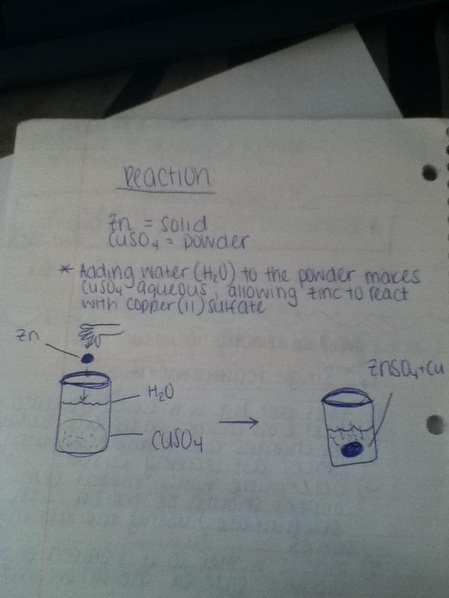
1. Measure the mass of the beaker
2. Then pour the CUSO4 in the beaker and measure the mass of the powder
3. Next fill the beaker up halfway with 25mL of water
4. Stir the water and CUSO4 until mixed together
5. Then take the ground up zinc and put into the beaker with the mixed water and CUSO4
6. Wait for a few seconds
7. In the next few seconds you should notice the Zinc reacting with the CuSO4 compound and water
8. You should have seen the color change from a bright blue to a dark blue color (shown above)
9. The copper should be at the bottom of the beaker returning to a copper color
10. When finished reacting Zinc should have combined with SO4 making Zinc Sulfate
*Adding water (H2O) to the powder makes CuSO4 an aqueous or soluble
solution. This allows Zinc to react with Copper (II) Sulfate making Zinc
Sulfate*
Conclusion
In this experiment I showed how to create the chemical Zinc Sulfate. Zinc Sulfate is produced using Zinc and Copper Sulfate. In the single replacement reaction I used the Activity Series to help me find the weaker metal. Since Zinc is a stronger metal than copper I paired Zinc with Sulfate on the reactants side of the equation. This makes Copper independent on the reactant side and also forms my chemical ZnSO4. After knowing this I proceeded to complete my lab experiment. In order to begin my lab I had to do some measuring. I had to measure the mass of the beaker, which came out to be 29 grams. I also had to measure the mass of the Zinc, which could only be measured inside the beaker. The Zinc and the beaker together weighed 34 grams. I then subtracted the total mass minus the beaker mass to find the mass of the Zinc alone. The Zinc weighed 5 grams. Next I began my experiment. Halfway through the lab I made a human error. The Zinc pebbles did not cause the CuSO4 to react, so I had to add extra Zinc, but this time in fine powder. This affected my measuring in the beginning of the experiment. This caused there to be more grams of Zinc in the beaker than neccessary.
Work Cited
1. Chemical Elements.David E. Newton. Ed. Kathleen J. Edgar. Vol. 3. 2nd ed. Detroit:
UXL, 2010. P673-681. From Gale VirtualReference Library.
2. Encyclopedia of Food and Culture. Ed. Solomon H. Katz. Vol. 3. New York: Charles Scribner's Sons,
2003. P409-410. From Gale Virtual Reference Library.
3. The Gale Encyclopedia of Medicine.
Vol. 3 3RD ed. Detroit: Gale, 2006. P2446-2448. From Gale Virtual reference library.
4. www.cdc.gov/niosh/ipcsneg/neng1698.html
5. www.dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=51710
6. www.teck.com/DocumentViewer.aspx?elementId=108520...tc